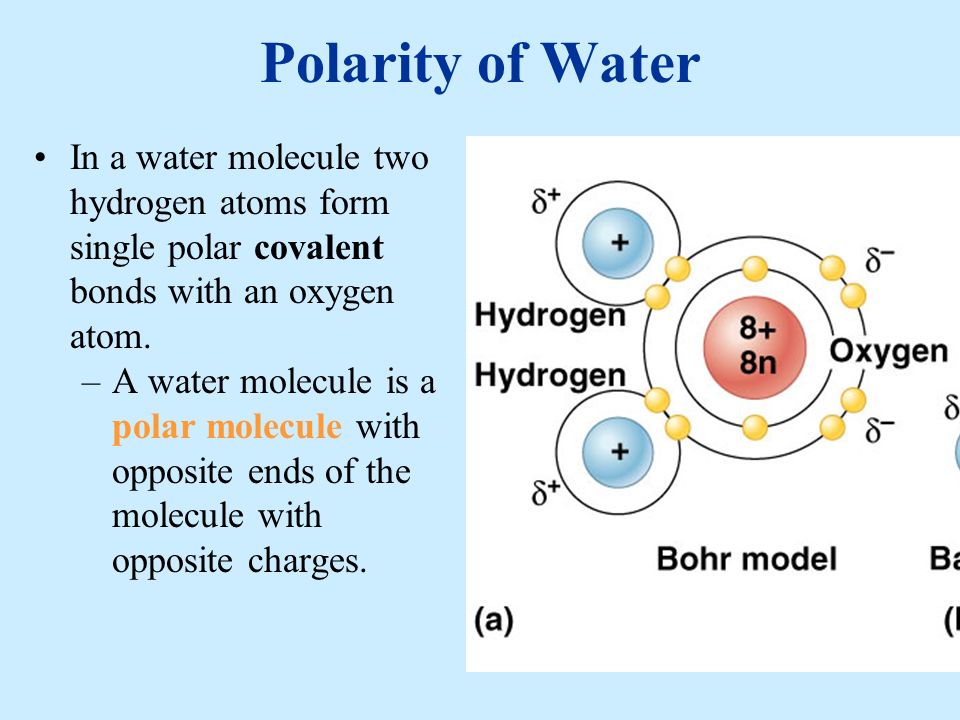
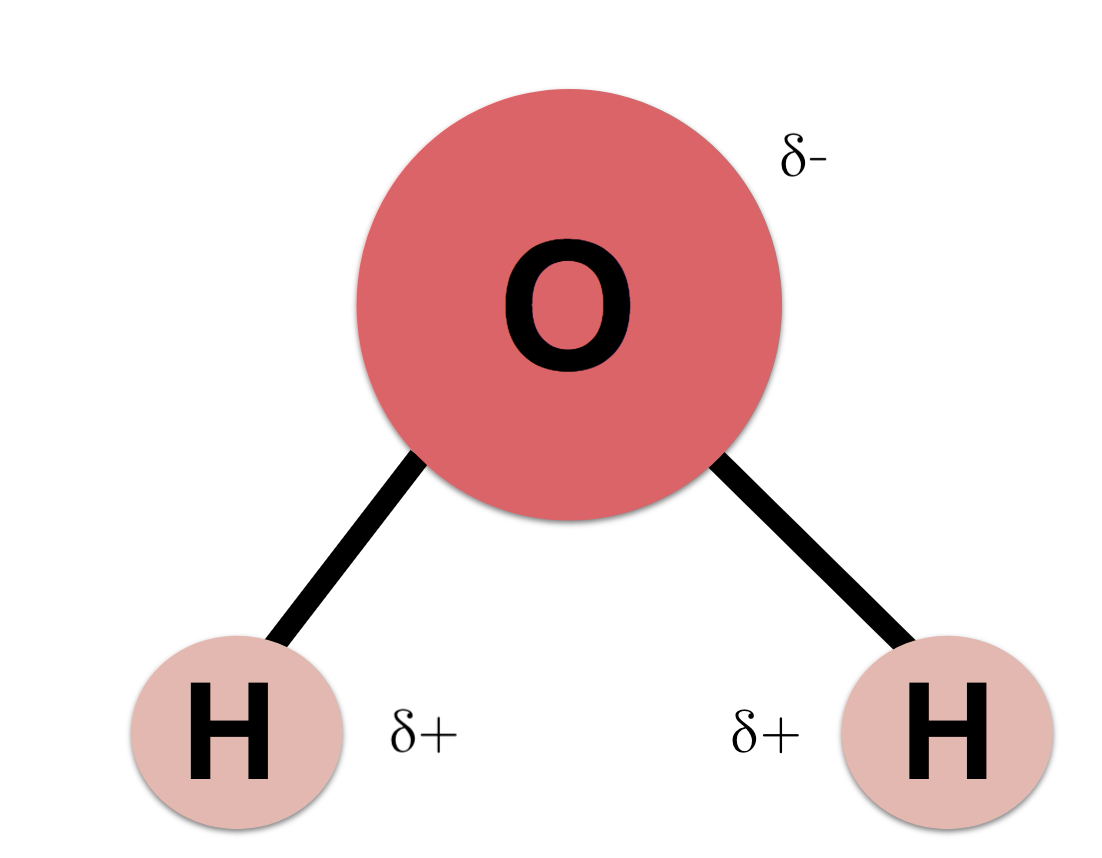
Describe the Polarity of a Water Molecule
Water is a tasteless odorless liquid at ambient temperature and pressureLiquid water has weak absorption bands at wavelengths of around 750 nm which cause it to appear to have a blue colour. There are millions of these molecules in one drop of water.

The Strong Polar Bond Between Water Molecules Creates Water Cohesion U S Geological Survey
Water H 2 O is polar because of the bent shape of the molecule.

. Describe the polarity of a water molecule. A water molecule is polar but it has no net charge. This is an example of polar covalent chemical bonding.
Polar molecules attract one another by dipole-dipole forces as the positive end of one molecule is attracted to the negative end of the nearby molecule. This can easily be observed in a. While there is no net charge to a water molecule the polarity of water creates a slightly positive charge on hydrogen and a slightly negative charge on oxygen contributing to waters properties of.
While there is no net charge to a water molecule the polarity of water creates a slightly positive charge on hydrogen and a slightly negative. The hydrogen and oxygen within water molecules H2O form polar covalent bonds. Water is a polar molecule which means it can attract other polar molecules.
Change the bond angle to see how shape affects polarity. See how the molecule behaves in an electric field. Change the electronegativity of atoms in a molecule to see how it affects polarity.
One molecule of water has two hydrogen atoms covalently bonded to a single oxygen atom. Thus water will adsorb to substances such as cellulose cotton but not to polyesters with few oxygen. A water molecule is formed by a combination of two hydrogen atoms and one oxygen atom.
The net result of these charges gives water its polarity. Hydrogen bonds form between multiple h2o molecules because each molecule is polar has to sides Each h20 molecule is polar because of a high electronegativity of oxygen and ther middle electronegativity of hydrogen. It is called H2O because it has two atoms of hydrogen H and one atom of oxygen O.
It can form hydrogen bonds with other elements. Covalent bonds in a water molecule bind its oxygen atom to its two hydrogen atoms A water molecule is polar but it has no net charge. Strong covalent bonds are formed between two or more water molecules.
Because of the higher electronegativity of the oxygen atom the bonds are polar covalent polar bonds. The shape means most of the negative charge from the oxygen on side of the molecule and the positive charge of the hydrogen atoms is on the other side of the molecule. This makes water an extremely potent solvent.
This causes on end of the molecule to be negative while the other is positive. The oxygen atom in a water molecule is strongly electronegative. The adhesion of water is explained by hydrogen bonding of water molecules to other polar surfaces.
A water molecule is polar because there is an uneven distribution of electrons between the oxygen and hydrogen atoms. The level of polarity in water is extremely high uniquely so. Water is polar due to the uneven sharing of electrons between oxygen and hydrogen.
When is a molecule polar. Structure of Water. The polarity of water can be calculated by finding the sum of the two dipole moments of both O H bonds.
Polarity of a Water Molecule. Covalent bonds in a water molecule bind its oxygen atom to. The scientific name for water is H2O.
Water molecules are polar and thus associate with each other through hydrogen bonds. μ Q r. A molecule is a piece of matter that contains two or more atoms.
The water molecule is very simple. Where μ is the dipole moment Q is the coulomb charge Q 160 1019C and r is the bond length or the distance between two ions. For ionic compounds the dipole moment could be calculated by.
The oxygen molecule is slightly negative and the hydrogen molecules are slightly positive. The oxygen atom attracts the shared electrons of the covalent bonds to a significantly greater extent than the hydrogen atoms. Water is the chemical substance with chemical formula H 2 O.
The outcome is a difference in charge or polarity from one end of the molecule to the other. Students will watch a video lesson discuss new information and apply new. The polarity of water is the net result of the overall negative charge of the oxygen atom and the overall positive charges of the two hydrogen atoms.
Water is a polar molecule and polarity occurs when the electrons in molecules are not spread evenly. Water is a simple molecule consisting of one oxygen atom bonded to two different hydrogen atoms. This creates a partial charge called a dipole.
Atoms are the tiniest pieces of matter. Water is a polar molecule as greater electron density is found around the more electronegative oxygen atom. The oxygen atom in a water molecule is strongly electronegative.
According to the State of Utah Division of Water Resources a water molecule is polar because the oxygen atom at the top of. The hydrogen atom bonds to each oxygen atom with a pair of shared electrons. An excellent example of the high polarity of water would be the fact that salt dissolves in water.
Waters Polarity One of waters important properties is that it is composed of polar molecules. The oxygen atom is more negatively charged at one end of the water molecule while the hydrogen atom at the other end is positively charged. Water has polar bonds and a linear symmetrical shape so it is polar.
In this lesson plan students will explore the concept of molecular polarity. Which statements accurately describe the polarity and electronegativity of water. Water molecules are attracted to each other to form hydrogen bond because because of multiple h20 molecules.
Many plants have hydrophobic coatings on their leaves. The strong dipole of water exerts electrostatic and gravitational forces on charged electrovalent compounds and on the dipoles of polar covalent compounds. Water has nonpolar bonds but it has a bent shape and is therefore polar.
Water Describe Water 46nbtop How Would You Describe Water To Someone Who Had Never Seen It Before You Might Say That Pure Water Has No Color No Taste Ppt Download
0 Response to "Describe the Polarity of a Water Molecule"
Post a Comment